The Persistence of Influenza
In this post, I am covering some topics related to influenza, including antigenic shift vs. drift, immunodominance, and original antigenic sin (OAS). These aren’t unique to influenza, but they have been well characterized in influenza, one of the most common human viruses.
Influenza Overview
There are 4 major influenza types – A, B, C, and D. Influenza A is the most common type, and its natural reservoir is birds. Influenza B is similar to type A but can only spread from human to human and accounts for approximately 25% of human influenza infections 1, while influenza A can be transmitted across species. Influenza C causes relatively mild infections and doesn’t cause epidemics, and influenza D only infects pigs and cattle. 2
Of the influenza types, influenza A is the biggest threat to humans and has caused several pandemics in the past century. Due to its rapid accumulation of mutations and reshuffling of genes, influenza A subtypes are named according to the nature of their surface proteins – hemagglutinin and neuraminidase. This gives rise to familiar names of pandemic influenza A viruses with the years in which they emerged, such as H1N1 (1918 and 2009), H2N2 (1957), and H3N2 (1968). These denote the type of hemagglutinin and neuraminidase in each pandemic influenza strain. The number doesn’t mean anything, just that H1 is different from H2, which is different from H3. Three hemagglutinin (H1, H2, and H3) and two neuraminidase (N1, N2) variants have been found in influenza A viruses that spread among humans. The following figure shows the major structural components of the influenza A virus.
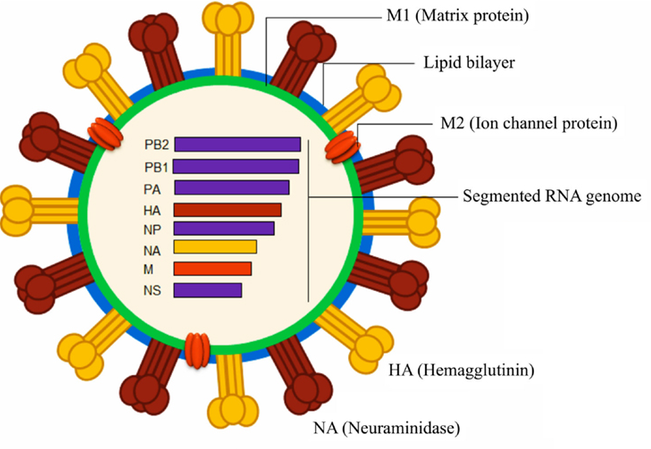
Hemagglutinin binds to host cells in order to infect them. It binds to a sugar group called sialic acid, which is found on the surface of many cell types. Neuraminidase allows newly made influenza viruses to leave a cell to go infect more host cells. When the viruses start to emerge from the cell, neuraminidase removes sialic acid groups from the surface so that influenza viruses don’t get stuck to the original cell (which will die). Neuraminidase inhibitors are commonly used influenza antivirals, such as Oseltamivir (Tamiflu), Zanamivir (Relenza), and Peramivir (Rapivab) 3.
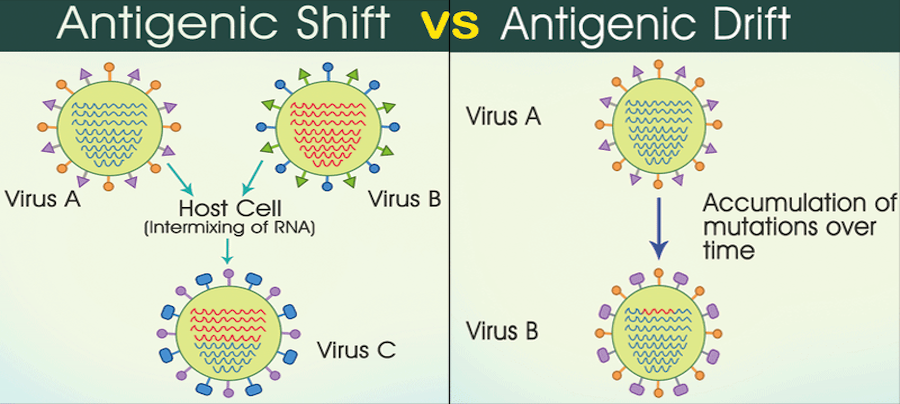
In the virus in the diagram above, we see that influenza has a segmented RNA genome. Its genome doesn’t consist of a single long piece of RNA, but is instead broken up into 8 segments (influenza C is the only one with 7 segments), with each segment encoding 1 or more influenza proteins. The segmented genome is particularly concerning for hosts because if multiple influenza strains infect the same cell simultaneously, they can rearrange themselves and form a virus with a new genome. This is analogous to how the genetic material of human parents is recombined to create a new human being, but more than 2 parent influenza strains can give rise to a new viral strain. Each segment could, in theory, come from a different parent strain.
This phenomenon is called antigenic shift. This is in contrast to antigenic drift, the process by which mutations in a virus change its genome and consequently its proteins. Antigenic shift can be thought of as large leaps in genetic diversity that occur through strains swapping their genetic material, while antigenic drift is generally a slower process driven by random mutations. This blog post has a good description of influenza genome reassortment. However, too many mutations can inactivate a virus because mutations inevitably disrupt an essential protein.
How RNA Viruses Acquire Diversity
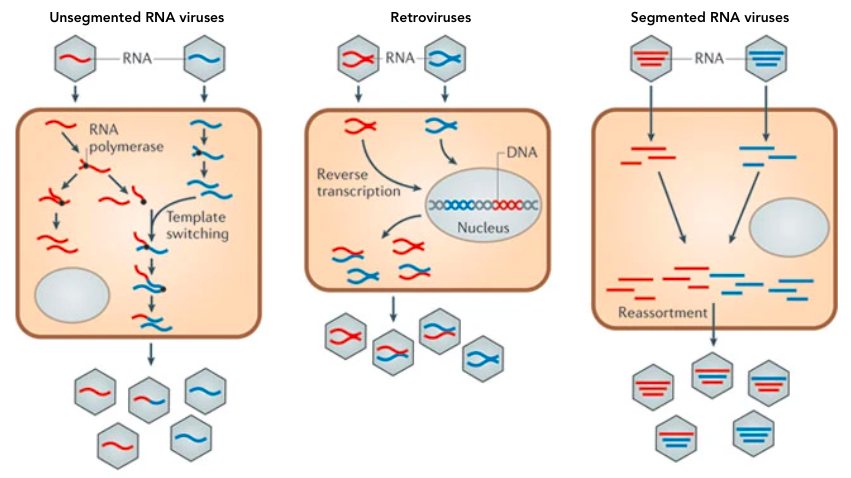
Upon infection by a virus, a host cell makes (often) identical copies of the virus, except for mutations that occur randomly to cause genetic variation. However, RNA viruses are particularly unstable and have other methods of shuffling their genetic material to generate more population diversity. Viral recombination can also occur when viral RNA is copied to make genomes to package into new viruses. The RNA-dependent RNA polymerase enzyme, which copies viral RNA, can sometimes switch between strands. For example, if multiple strains of a virus infect a single host cell and release their RNA into the cytoplasm, the viral polymerases can copy one part of the genome from one strain and the other parts from other strains. This is aptly called template switching and typically occurs in viruses with non-segmented (a.k.a continuous) genomes.
Another recombination mechanism is specific to retroviruses, RNA viruses like HIV that reverse transcribe their RNA into DNA and integrate it into a host’s genome. This makes retroviral infections difficult to clear because once the viral genome is part of the host’s DNA, it will be replicated along with the host’s genome and can re-start an infection at any time. Retroviral recombination occurs if a single host cell has been infected with multiple strains of the same type of retrovirus. The following figure comes from a paper titled “Why do RNA Viruses Recombine” by Etienne Simon-Loriere & Edward C. Holmes to demonstrate the different RNA virus recombination routes 4.
Influenza Antibodies
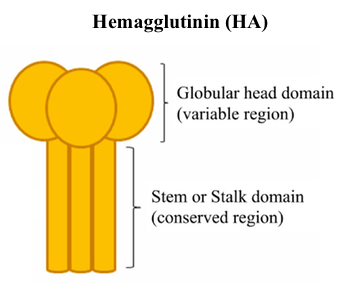 The hemagglutinin (HA) surface protein has two primary regions -- the head and the stem. The head domain has more variation across influenza A isolates in different people, whereas the stem domain is more similar. This means that anti-head antibodies will be more specific and therefore less likely to protect you from getting reinfected with other influenza strains. Anti-stem antibodies are more likely to provide broad protection because they bind to the same region.
The hemagglutinin (HA) surface protein has two primary regions -- the head and the stem. The head domain has more variation across influenza A isolates in different people, whereas the stem domain is more similar. This means that anti-head antibodies will be more specific and therefore less likely to protect you from getting reinfected with other influenza strains. Anti-stem antibodies are more likely to provide broad protection because they bind to the same region. A single pathogen contains many proteins, each of which is an antigen, something that can be bound by an immune receptor. Proteins are typically 100-1000 amino acids long, with variation across eukaryotes, prokaryotes, and viruses. Within each antigen, there are numerous epitopes, the exact sequence to which an antibody or lymphocyte binds. An epitope is usually 5-10 amino acids long and includes both the sequence and its 3D conformation in space.
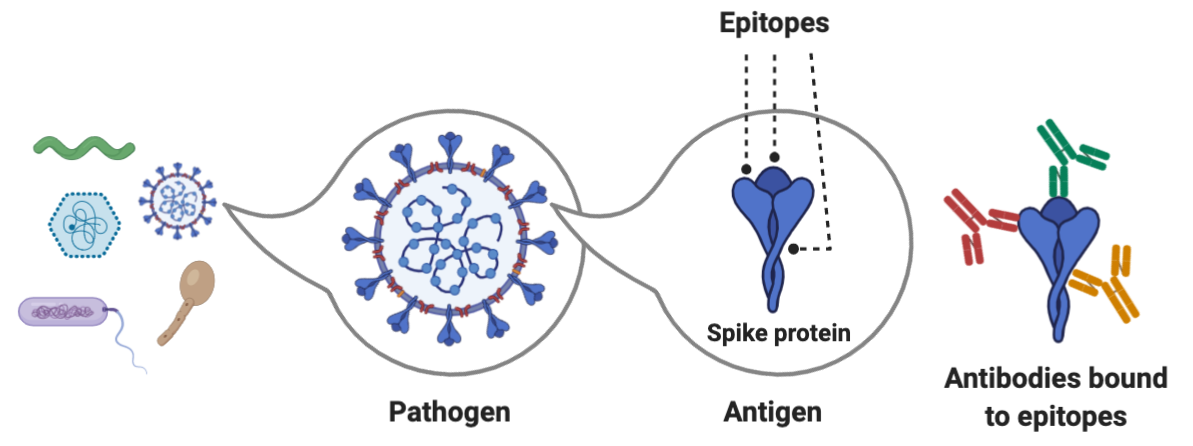
The head of hemagglutinin is also immunodominant to the stem, eliciting a greater immune response. During lymphocyte activation, some B and T cells proliferate to produce memory cells, which stay in the blood and lymph nodes after the infection has been cleared. A balance is needed between having many memory cells for a single epitope (to maximize sensitivity and the strength of the memory response) and a diverse set of memory cells, which maximizes epitope coverage, but can lead to fewer cells for each epitope. Some epitopes get favored through immunodominance and their antibodies and memory cells are overrepresented.
One hypothesis is that large differences in affinity and avidity of B cell receptors (BCRs) on naive B cells skews an immune response. In terms of antibodies, for which this concept is most significant, affinity is the strength of binding between a single antibody and its antigen, and avidity is the total strength of binding when many antibodies bind to an antigen simultaneously. Often, one interaction increases the likelihood that more antibody:antigen interactions will occur because proteins are brought close together through clustering. The hypothesis for influenza is that some BCRs have much lower affinity for the virus and therefore get outcompeted. B cells that bind tighter to the virus are selected for, and they proliferate. Another possibility is that some BCRs have a harder time binding to their epitopes on the surface of influenza, so even if they bind with high affinity, their epitopes are not as accessible as others. 5. Still a third hypothesis is that glycosylation sites, carbohydrate chains attached to certain amino acids, of hemagglutinin could cover up certain epitopes and make others dominant 1.
This provides interesting insight into developing better influenza vaccines. We currently use inactivated flu vaccines – formaldehyde-treated influenza strains – but a live-attenuated flu vaccine (given intranasally) is also available. These are both quadrivalent, containing H1N1, H3N2, and the two strains of influenza B, while the previous trivalent vaccine contained only one strain of influenza B. Because of antigenic drift, the flu vaccine must be updated each year based on predictions of the most common antigenic changes. These predictions are made by the World Health Organization 6 months in advance of the next flu season, but countries manufacture their own vaccines. Currently, the flu vaccine is effective at preventing around 40-70% of flu cases, depending on how similar the vaccine is to the circulating strains and characteristics of individuals, such as age, health conditions, and pregnancy 6.
Flu vaccines that contain only the stem domain could be a way to induce immunity against the more conserved stem of HA. This would be more protective against influenza strains because the stem doesn’t change as much as the head 7. In immunization of mice with only the stem of HA, a robust antibody response was observed against the stem, suggesting that stem subdominance is caused by its being attached to the head 8. Furthermore, when mice were immunized with both stem proteins and full HA, fewer stem-specific B cells were observed. This indicates that there might be competition between B cells specific to the two types of proteins for activation.
Original Antigenic Sin
Original antigenic sin (OAS), also known as antigenic seniority or antigenic imprinting, describes the phenomenon that the first strain that you are exposed to skews the memory responses to future infections by the same virus. This is most prevalent in influenza, and the reasons are not well understood. This term was coined by American microbiologist Thomas Francis Jr. in 1960 based on evidence that the influenza antibodies present at highest concentrations in people were for strains they had been exposed to during childhood. Even though people had contracted influenza later in life, the earliest strains encountered seemed to stimulate the strongest antibody response and is seen as the most important influenza exposure in a hierarchy of strains in the body 9,10.
This phenomenon only occurs between viruses that are sufficiently related, so immunity against an influenza strain will not skew your immunity to something like a cold-causing virus and vice versa 9. When you get infected by strains of influenza that are different from the first strain you contracted, the immune response is not as specific to the new strain. It’s as if the memory of the first infection gets reactivated, which prevents you from learning how to respond to the new strain.
In the figure below, the first influenza strain binds to a naive T cell and stimulates an immune response, and long-lived memory cells circulate in the blood in case of reinfection. However, if a slightly different strain of influenza (a strain with different looking proteins) infects the same person, then it can bind to the memory cells from the first infection, rather than binding to naive T cells. There is some degree of cross-reactivity between influenza strains that prompts a memory response from a past infection. In this way, a suboptimal immune response is raised instead of one more specific to the new influenza.
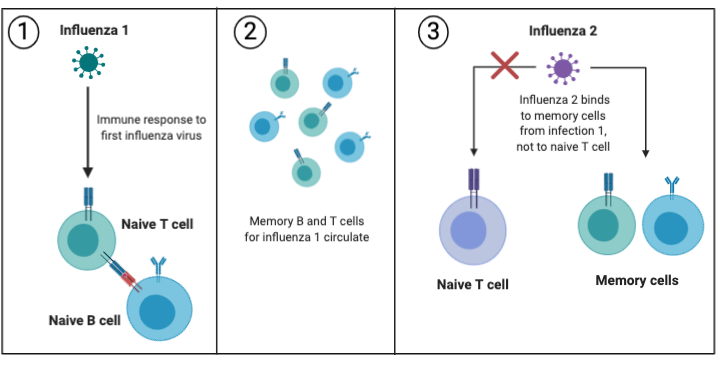
Pandemic Influenza
Original antigenic sin has been proposed as an explanation for the unexpected mortality curve observed in the 1918-1919 influenza pandemic. This pandemic was notorious for being deadly for individuals 20-40 years old, who normally fare better than other age groups. In a study from 2001, the authors determined the excess mortality rate for each age group, comparing the numbers of deaths during the three influenza pandemics in the 20th century to the numbers of deaths in the 5 years before each pandemic 11. The data are shown below:
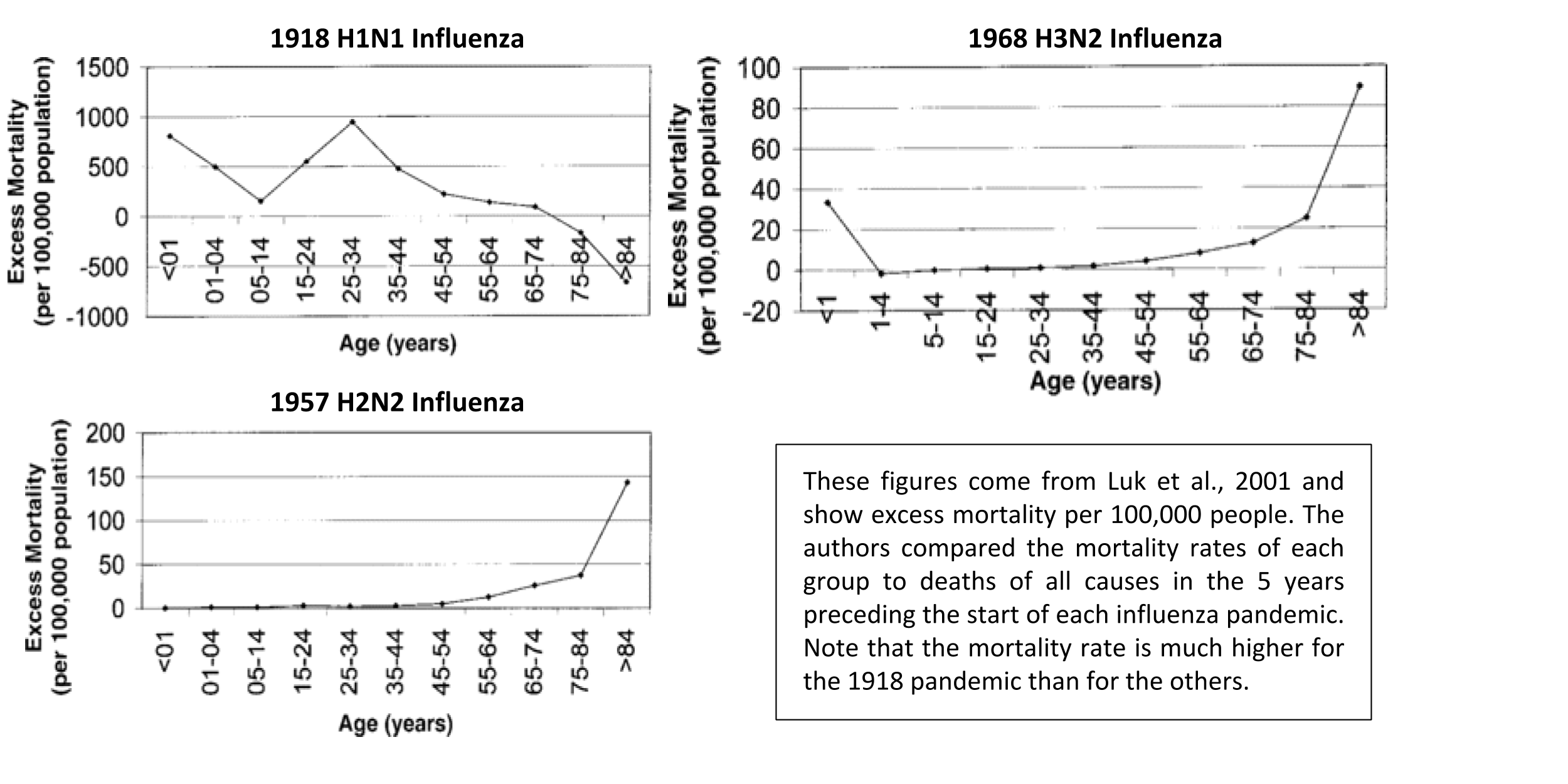
For most diseases, peaks in mortality occur in the very young and the very old because they have weaker immune systems and more health conditions. In the 1918 pandemic, a peak in excess deaths occurs in the 25-34 age range (specifically at age 28), which is anomalous. A paper from 2014 proposes that exposure to a certain influenza strain in the 1889-1890 season in infancy caused a suboptimal immune reaction to the 1918 H1N1 strain in these same people 12. Previous sero-archaeological (“serum” = antibody) studies found higher antibodies for H3 and N8 in people born in and around 1889, suggesting that an H3N8 influenza strain was circulating at that time and was the first influenza exposure for infants.
The authors of the 2014 paper estimated that people who were born before or after 1889-1890 had their first exposure to influenza strains with H1 or N1 more similar to the 1918 virus, which may have put the 1889-1890 cohort at a disadvantage because of antigenic imprinting 12. Similarly, during the 2009 H1N1 pandemic, which caused 491,000 cases, children had very high rates of influenza infection and hospitalization, and younger adults died at a higher rate than the elderly 13,14, which is not characteristic of seasonal influenza, but appears to be common among pandemic strains. In one study, older adults were found to have more cross-reactive antibodies against the 2009 H1N1 strain 15, and it is hypothesized that these antibodies came from childhood exposure to similar strains.
Pandemic influenza is worrisome because it seems to emerge and disappear rather abruptly. This may be explained by influenza being a zoonotic virus that originated in birds, which are its natural reservoir, and its ability to reshuffle its segmented genome across strains. Since influenza is carried by many animals – primarily birds, pigs, and humans – there are many opportunities for mixing of strains. Genes for proteins can be swapped between strains from different animals, and so new combinations can arise. Influenza strains can therefore seem to “hide” from humans and emerge suddenly, but they’re simply circulating in other animals. They appear when transmitted directly from animals to humans or when strain reassortment occurring during co-infection of a single animal by multiple influenza strains brings new proteins to humans. Regardless of what animal they’re in, influenza strains undergo antigenic drift. While a certain protein may disappear from humans and then reappear again at a later time, the mutations its associated gene acquires make it distinct from before. However, it is still genetically similar, allowing scientists to track influenza evolution and emergence.
When a new variant of an influenza protein that most or all people have not yet encountered emerges, the strain harboring this protein has pandemic potential. This is because there is very little preexisting immunity in the human population, so the virus can spread throughout the population if given the chance. In such situations, scientists can study the mortality rates and antibody responses to determine if preexisting immunity helps or harms people, as was speculated to have occurred in the 1918 pandemic. Differing accounts of whether OAS is helpful or harmful complicate the matter, and sequential immunizations are difficult to study in a laboratory setting as they would occur in humans.
Conclusion
Influenza is a difficult virus to pin down. With its segmented genome and fast mutation rate, it is highly changeable. Since the head of hemagglutinin (HA) is immunodominant to the stem, a stronger response is raised against the varying head. But this reduces protection against other influenza strains because they likely have a different head region. Many scientists are working towards a universal flu vaccine, one that may target the stem of hemagglutinin or another conserved protein, such as M1, M2, and the nucleoprotein 16. Some adjuvants 17 and novel vaccine delivery methods 8 may aid in the induction of stem-specific antibodies. Many of these have shown promise in animal models and are in early clinical trials.
Leave a Comment