Introduction to the Science of COVID-19
This post is an overview of the science of COVID-19 based on information from scientific papers, government agencies, and media outlets. In future posts, I will discuss in more detail topics like vaccines/treatments, zoonotic virus spillovers, viral evolution, SARS-CoV-2 in the context of other viruses, and demographics of patients.
What Are Viruses?
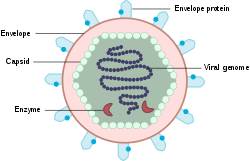
The basic structure of viruses is genetic material (DNA or RNA) and maybe some enzymes enclosed in a protein coat called the capsid. Some viruses, including coronaviruses, have a lipid bilayer that surrounds the capsid called an envelope. The envelope is studded with glycoproteins, which bind to host cell receptors.
To enter a cell, a virus begins by binding to receptors on a cell. The surface of non-enveloped (or “naked”) viruses does not interact favorably with the phospholipids that make up the cell membrane, so these viruses typically enter through vesicles -- small enclosed compartments that carry materials inside a cell. This process is called receptor-mediated endocytosis because it brings particles inside (“endo”) a cell (“cyto”).
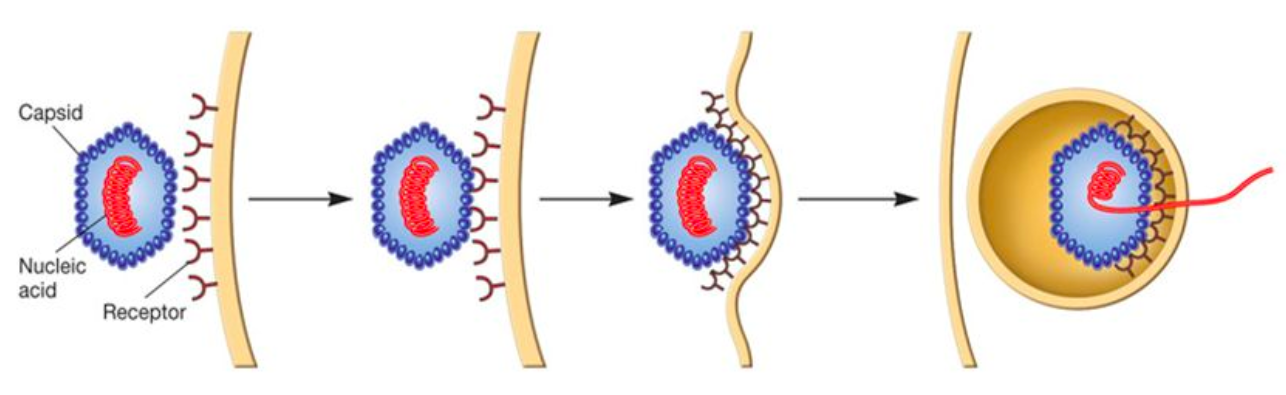
After endocytosis of a virus, the vesicle fuses with another type of cellular compartment called an endosome. The endosome is an acidic (low pH) environment, which is thought to prepare the virus before it is released into the cytoplasm. Enveloped viruses can also use receptor-mediated endocytosis, but since they are covered by the same phospholipid bilayer as the cell membrane, they can also fuse with host cells and directly release their capsids.
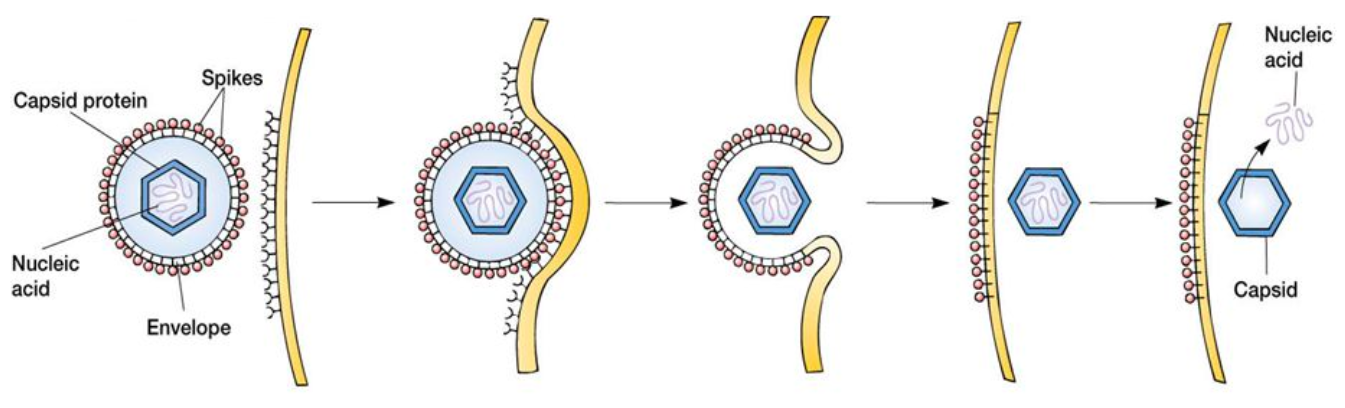
Once viruses are released into the cytoplasm, they are uncoated to free their genetic material. The next step varies between DNA and RNA viruses, but the host cell’s protein production machinery is used to produce more viruses. Once the new viruses are assembled, the infected cell typically bursts, releasing its viruses to go infect more cells.
Viruses mutate alarmingly quickly. Higher eukaryotic organisms, such as humans, contain many enzymes that proofread our genomes to correct mistakes. Viruses typically do not contain these proofreading enzymes, and therefore, their genomes change much faster, allowing them to acquire new traits and mutate away from our immune systems or treatments. Most mutations in viruses are deleterious because they change an essential protein, but some can be advantageous.
The World of Coronaviruses (CoV)
Coronaviruses are enveloped RNA viruses, so named because their spike proteins form a crown (corona) around the surface 1. Seven coronaviruses have been identified that infect humans, four of which only cause cold-like symptoms 2. The other three, which are more severe, are SARS-CoV (Severe Acute Respiratory Syndrome), MERS-CoV (Middle East Respiratory Syndrome), and SARS-CoV-2, which causes COVID-19 (COronaVIrus Disease 2019) 2.
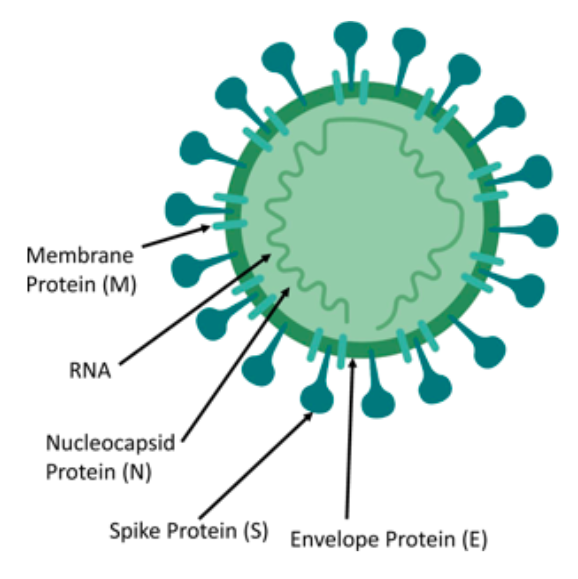
Left: A coronavirus with the primary components. Viruses are minimalistic, and they contain as few proteins as possible to achieve their goals. The spike protein interacts with host receptors and facilitates viral entry. Coronaviruses also have other proteins -- membrane, envelope, and nucleocapsid -- that are involved in viral structure, assembly, and RNA binding.
The SARS outbreak began in late 2002 and lasted until July 2003, during which there were 8,098 cases and 774 deaths 3. MERS was first identified in September 2012, but cases have emerged periodically owing to its continued circulation in camels. Since then, there have been 2,494 cases and 858 deaths 4. The outbreaks of SARS and MERS did not become pandemics due to their larger fatality rates and lower transmission rates relative to COVID-19. The fatality rates of SARS and MERS are ~10% and 30-40%, respectively, while the fatality rate of COVID-19 appears to be less than 5%.
SARS-CoV-2 spreads more easily than SARS-CoV and MERS-CoV because it remains in the nose and mouth longer. It also causes symptoms AFTER the period of maximum infectivity or even causes asymptomatic cases, making SARS-CoV-2 more difficult to contain than the other two. SARS-CoV and SARS-CoV-2 bind to the same receptor, and there is some evidence that SARS-CoV-2 may bind to it with higher affinity, which might mean that a smaller viral load could cause illness 5,6. RNA viruses mutate faster than DNA viruses, but coronaviruses, which have some of the largest RNA genomes among viruses, appear to have proofreading enzymes called exonucleases. When a virus can correct its own genomic mistakes, fewer lethal mutations occur, and the virus persists longer. 7.
Mechanism of Infection of SARS-CoV-2
SARS-CoV and SARS-CoV-2 have significant similarities: they have 70% total amino acid similarity and 76% spike protein sequence similarity and have caused similar disease symptoms. Consequently, current research on SARS-CoV-2 is using knowledge about SARS-CoV as a starting point for inquiry. The spike protein on the surfaces of SARS-CoV and SARS-CoV-2 bind to angiotensin-converting enzyme 2 (ACE2) 8. ACE2 is a protein primarily expressed by epithelial cells in the lungs, heart, kidneys, and small intestine, and it is part of the renin-angiotensin system (RAS), which regulates blood pressure 9.
SARS-CoV-2 utilizes at least two enzymes -- transmembrane serine protease 2 (TMPRSS2) and cathepsins -- to assist with cell entry. These enzymes are proteases, whose function is to cleave proteins at specific sites 10. TMPRSS2 is embedded in host cell membranes, and cathepsins are activated in acidic (low pH) endosomes to degrade pathogens after entry. Both proteases are involved in priming the spike proteins of SARS-CoV and SARS-CoV-2 before entry, and inhibiting only one enzyme may not fully protect against infection 10. Antimalarial agents, such as chloroquine and hydroxychloroquine, raise the pH of endosomes, so they may work against SARS-CoV-2 by inactivating cathepsins, but TMPRSS2 may still allow infection. A cocktail of drugs could be more effective by blocking multiple enzymes simultaneously.
SARS-CoV-2 contains a cleavage site for another protease called furin, which is made by our bodies and cleaves certain proteins to activate them 11. In many coronaviruses, the spike protein is cleaved by a furin-like protease, which assists pathogenesis. Furin-like cleavage promotes later activation of the spike protein of MERS-CoV 12, and proteases may also help viruses from animals infect humans 13. Similarly, furin might increase the pathogenicity of SARS-CoV-2, but this has not yet been studied.
Pathogenesis of COVID-19
Approximately 80% of COVID-19 cases are mild, but the symptoms are unpleasant. A mild case of COVID-19 is like the flu -- fever, fatigue, and a dry cough 14. Nasal congestion and a runny nose are less common, but there have been reports of diarrhea or vomiting in some severe cases. All tissues that express ACE2 are at risk of infection by SARS-CoV-2.
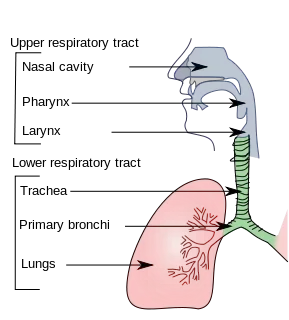
The difference between mild and severe cases depends on how far down in the respiratory tract SARS-CoV-2 is able to get a foothold. Mild cases occur when the virus remains in the upper respiratory tract -- nose and throat -- because there is no impairment of breathing, but when it infects cells in the lungs, pneumonia and respiratory failure can occur.
The primary causes of death from COVID-19 are viral pneumonia and respiratory failure. Viral pneumonia can lead to widespread lung damage, impairing breathing (acute respiratory distress syndrome) or causing multiple organ failure and septic shock. When the alveoli -- tissue sacs responsible for gas exchange -- deep inside the lungs become infected, they fill with pus and can not provide oxygen to the rest of the body as efficiently, leading to pneumonia. If large numbers of epithelial cells lining the respiratory tract die after becoming infected, they can no longer prevent other pathogens from getting into the lungs, leading to secondary infections. To protect against secondary bacterial infections, many hospitalized COVID-19 patients are given antibiotics, but antibiotics do not affect SARS-CoV-2 itself.
Sepsis is a condition of extreme inflammatory response. When the body is fighting an infection, the innate immune system responds first, attacking all pathogens indiscriminately. Signaling molecules called cytokines and chemokines cause inflammation, and immune cells attack pathogens and infected host cells. When inflammatory cytokines are produced, they activate more immune cells to produce even more cytokines, leading to an inflammatory cascade called cytokine storm syndrome. The widespread inflammation can damage a patient’s own cells, and patients may suffer organ failure due to lack of oxygen and extremely low blood pressure.
What is a Coronavirus Test?
A coronavirus test is a polymerase chain reaction (PCR) -- a laboratory technique that makes many copies of target DNA or RNA through repeated cycling. The technique is more specifically known as Real-Time Reverse Transcriptase PCR because it converts the SARS-CoV-2 RNA into DNA using an enzyme called reverse transcriptase before copying certain sequences that we know are present in the SARS-CoV-2 genome. The SARS-CoV-2 genome was released in early January 2020, and from that, probes -- small single-stranded DNA sequences that bind to specific locations in SARS-CoV-2 genome -- have been constructed. The probes are bound to fluorescent proteins, and as the reaction duplicates the target sequence in each cycle, the fluorescence can be measured to back calculate how much viral RNA was originally present.
Zoonoses and Spillover Events
Coronaviruses are zoonotic viruses, meaning they normally circulate in animal populations and are then transmitted to humans during a spillover event. Both SARS and MERS originated in bats and jumped to humans via an intermediate animal -- civets for SARS and camels for MERS. Both of these animals are in close contact with humans in certain parts of the world, and viruses have evolved clever ways to survive in multiple species 15.
The closest relative of SARS-CoV-2 is a virus called RaTG13 found in bats in 2013. Therefore, it is suspected that SARS-CoV-2 originated in bats, but we do not yet know how it got to humans. The Huanan Seafood Market in Wuhan, Hubei, China has been implicated because the first COVID-19 cases diagnosed in December 2019 were all in people who had connections to the market. However, it is unclear whether the virus jumped from bats to an intermediate animal at the market or if someone contracted the virus elsewhere and began spreading it at the market. At wildlife and wet markets, numerous animals are in close proximity with each other and with humans, making it easy for viruses to spread and mix 16. Genetic tracing can potentially determine the virus’ origins (see Trevor Bedford’s Nextstrain analysis for phylogenetic tracing).
Some Pitfalls of COVID-19 Information
It is difficult to pinpoint the death rate of COVID-19 while the pandemic is ongoing. Fatality rates are highly susceptible to testing ability, access and quality of health care, and human demographics, which vary wildly across the globe. SARS and MERS outbreaks in the past 2 decades stimulated some hard-hit countries to prepare for future outbreaks. South Korea put efficient and rigorous testing procedures in place after a 2015 MERS outbreak, and this increased preparedness in some countries will reduce their fatality rates 17. Many people are not getting tested because of a shortage of SARS-CoV-2 test kits -- some people present with symptoms, but if they are deemed “low-risk”, they may simply be sent home. Antibody tests, like some developed at Mount Sinai hospital in New York, NY, may be able to detect people who recovered from COVID-19, but never got tested 18. These are binding assays that test if antibodies in a patient bind to the spike protein of SARS-CoV-2 19.
Miracle drugs are unlikely to exist because viruses are very good at escaping human interventions, and SARS-CoV-2 has already proven to be a formidable foe. Blocking interactions between the SARS-CoV-2 spike protein and ACE2 may prevent the virus from entering cells, but eliminating ACE2 function may worsen lung damage from pneumonia 20. Many people take drugs called ACE inhibitors or angiotensin receptor blockers (ARBs), which target other parts of the RAS pathway, not ACE2. These medications may unintentionally increase ACE2 expression, but there is not enough evidence to say that they increase COVID-19 risk. In fact, since they help lower blood pressure and the risk of heart failure, stopping them could be worse for COVID-19 patients.
Since we are in the thick of the pandemic, many results about potentially effective drugs come from very small groups of patients (on the order of < 50 patients). But to determine if a drug is safe and effective to administer on a large scale, it must be tested in comprehensive randomized clinical trials. It would be dangerous to administer treatments without first examining all the effects, and in the early stages, it is difficult to distinguish between an effective treatment and the immune system successfully fighting off the virus on its own.
Leave a Comment